ARO3:5®
Unique in its composition with extracts from the three wild Aronia species and big-fruited Aronia.
Documented in clinical trials at Danish hospital.
Published results
We have just published the results from the Clinical trial “SUND” on the health effects of our Aronia composition ARO3:5® in Journal of Clinical Medicine. The clinical trial had 109 male participants and has been a collaboration between a range of MD´s, Professors and scientists from the regional Hopital Horsens, Odense University Hospital, University of Southern Denmark, Chr. Hansen A/S and Byrial ApS.
The trial design is a so-called golden standard design performed as a cross-over, double blinded, randomized study. This means that all participants over a period of 270 days have received both placebo and Aronia (blinded) capsules in two 90 days treatment periods separated by a 90 days washout period. Due to the EU rules we are not allowed to show the results from the study at our home page until we get a formal permission from EFSA. This application process may take a year or so from now, but you are welcome to search for the paper which is available online for free via Journal of Clinical Medicine.
Developed over the last 10 years
Our Aronia powder composition, ARO3:5®, has been developed over the last 10 years by Byrial ApS and comprise the bioactive compounds from the three wild species of Aronia mixed with the big-fruited Aronia.
Virinia, Vikilia, Swecia & Roar
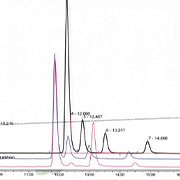
This mixture, ARO3:5®, is standardized according to the content of a total anthocyanins and comprise the three wild Aronia spp: Aronia arbutifolia var. Virinia, Aronia prunifolia var. Vikilia, and Aronia melanocarpa var. Swecia and the cultivated Aronia hybrid × Sorbaronia mitschurinii var Roar all developed via 18.000 controlled crossings at at Byrial Aps´s laboratory and research farm in Lejre Denmark.
Futher information
Please contact Henrik Byrial ( henrik@byrial.dk +45 22 61 10 66) for further information